The Phase II Clinical Trial of the Recombinant Protein COVID-19 Vaccine Kicks off

Open the phone and scan
It is reported that the phase II clinical trial of the recombinant protein COVID-19 vaccine (Sf9 cell) from West China Hospital of Sichuan University/Chengdu WestVac Biopharma Co., Ltd. was launched in Sheyang County, Jiangsu Province on the morning of November 18 by accepting volunteers for enrollment and injection. The research team, headed by Prof. Zhu Fengcai, a renowned vaccine expert from Jiangsu Provincial Center for Disease Control and Prevention (CDC), included local hospital physicians, research nurses and authorized medical staff from the testing department and GCP-related departments of Sheyang CDC. The Phase II study of the recombinant protein COVID-19 vaccine (Sf9 cell), as a single-center, randomized, double-blind, placebo-controlled clinical trial, is planned to enroll 960 individuals to evaluate the immunogenicity and safety of different doses of the recombinant protein COVID-19 vaccine administered by different immunization procedures in people aged 18 years and older.
The kick-off meeting and training were organized locally and attended by leaders of the Standing Committee of Sheyang Party Committee, Sheyang Health Commission, Sheyang CDC and relevant government organs. Academician Wei Yuquan, Director of the State Key Laboratory of Biotherapy at West China Hospital, Sichuan University, Zhu Fengcai, Deputy Director of Jiangsu Provincial Center for Disease Control and Prevention, and Wang Zhenling, a research fellow of the State Key Laboratory of Biotherapy at West China Hospital, Sichuan University attended the kick-off meeting.
Chengdu WestVac Biopharma Co., Ltd. (“WestVac”) is a biopharmaceutical company specializing in vaccine R&D and production and immunotherapy, which was established by the research team led by West China Hospital and Academician Wei Yuquan to develop the recombinant protein COVID-19 vaccine. Just two days ago, the company held a signing ceremony with Sichuan Development Holding Co., Ltd. and Shanghai Pharmaceuticals Holding Co., Ltd. for raising nearly 300 million yuan in its series A financing. The company is now building a new vaccine production line with an annual capacity of hundreds of millions of vaccine doses in Chengdu High-tech Zone. Insect cells have previously been used to develop recombinant protein vaccines against cervical cancer and influenza in Europe and the United States, and the approach has been tested to be safe and effective for protective immunity on humans. Besides producing COVID-19 vaccines with insect cells, WestVac Biopharma is also making rabies vaccines, influenza vaccines and herpes virus vaccines with the insect cell technology, as well as developing 20 odd innovative products for immunotherapy (including several tumor vaccines), with over 30 patents for related products and technologies obtained or applied.

Group photo at the kick-off meeting and training session

Group photo at the clinical trial site

Academician Wei Yuquan and Prof. Zhu Fengcai carry out research on site
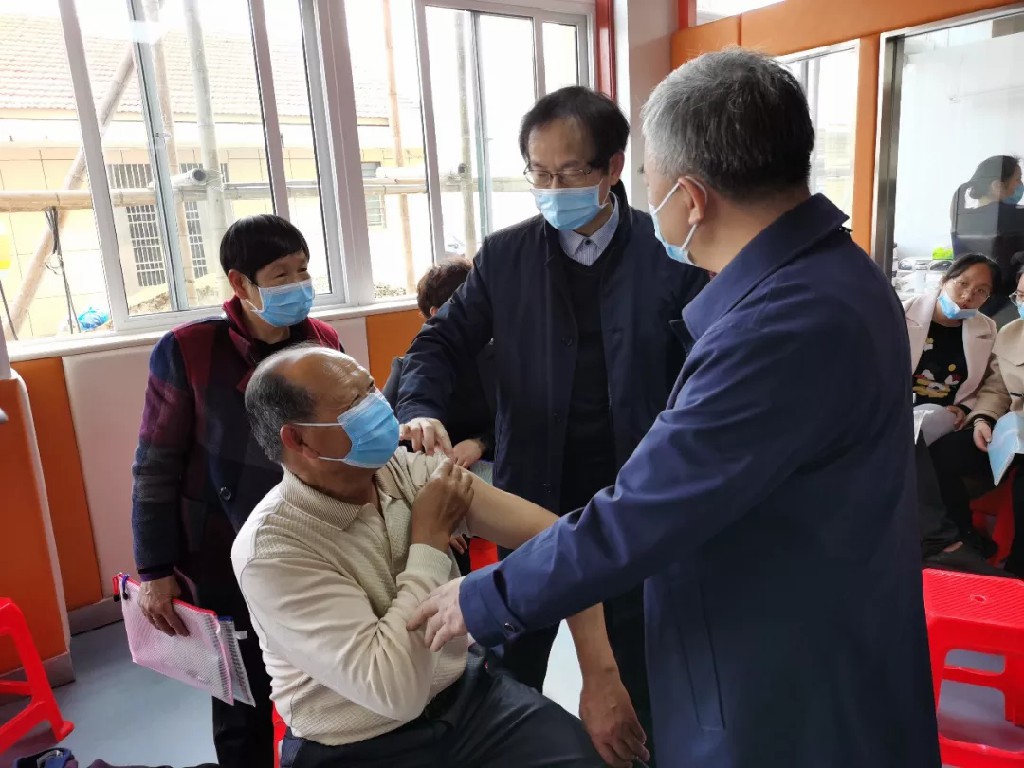


Academician Wei Yuquan and Prof. Zhu Fengcai observe subjects together


